  
Mouse hybridoma cell line 5E4/1F1 was kindly provided by Miha Kosmač and Vladka Čurin Šerbec (University of Ljubljana). Both cell lines were deposited to the DSHB by Kazumasa Takeda and Asako Sugimoto (DSHB hybridoma products KT13 and KT22). Mouse hybridoma cell lines KT13 and KT22 were obtained from the Developmental Studies Hybridoma Bank (DSHB). The HEK 293T cell line was obtained from the American Type Culture Collection (ATCC CRL-3216). This method makes paired Ig sequencing widely applicable even for laboratories without specialized equipment and personnel. Here, we describe a high-throughput method which enables sequencing of paired HC-LC immunoglobulin (Ig) repertoires from millions of B cells simply by using a cooled table-top centrifuge, a magnetic stirrer, and a thermal cycler. Although substantial, the existing methods are limited by their commercial availability, high costs, and require an elaborate construction of flow-focusing or microfluidic devices and dedicated personnel for operation. More recently, two emulsion-based methods reported paired HC-LC repertoire sequencing from 2–3 × 10 6 B cells at single-cell level. To this end, several single-cell paired sequencing technologies were reported more recently, which were initially limited by low cell numbers (< 400–10 5 cells) and sometimes required the use of complex microfluidic systems however, cellular throughput is improving through newer developments, such as droplet-based systems and the 10× Genomics platform. Retaining paired HC-LC data from bulk B cell populations at single-cell level remained a major obstacle for a long time. Initially, antibody repertoire analysis focused on obtaining information from antibody heavy chains (HC) only, missing the native light-chain (LC) pairing information that is necessary for antibody cloning and expression. 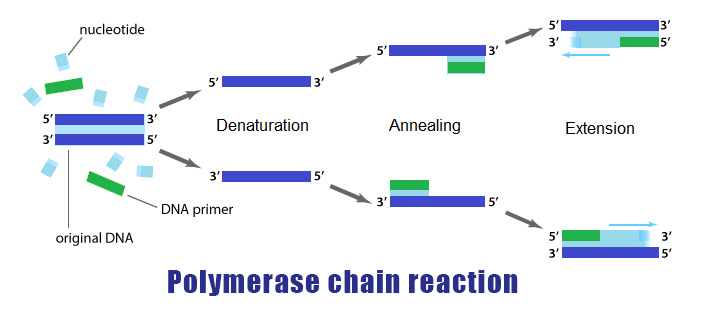
doi: 10.1016/j.foodres.2013.01.031.High-throughput sequencing of immunoglobulin repertoires from B cells has emerged as a powerful tool to investigate repertoire changes for antibody discovery, vaccine efficacy studies, and in other healthcare applications. Ecology of moulds during the pre-ripening and ripening of San Daniele dry cured ham. Description of the microflora of sourdoughs by culture-dependent and culture-independent methods. Iacumin L., Cecchini F., Manzano M., Osualdini M., Boscolo D., Orlic S., Comi G. Microbial, chemico-physical and volatile aromatic compounds characterization of Pitina PGI, a peculiar sausage-like product of North East Italy. Iacumin L., Osualdini M., Bovolenta S., Boscolo D., Chiesa L., Panseri S., Comi G. PCR-DGGE fingerprints of microbial succession during a manufacture of traditional water buffalo mozzarella cheese. Profiling of complex microbial populations by denaturing gradient gel electrophoresis of polymerase chain reaction-amplified genes coding for 16S rRNA. Muyzer G., de Waal E.C., Uitterlinden A.G. Panel B: PCR, conventional PCR ePCR, emulsion PCR NEC, non-emulsified control 10 −2, 10 −3, 10 −4, 10 −5, 10 −6, sample dilution on MSA agar at which the colonies were harvested in bulk and subjected to total DNA extraction and PCR/ePCR analysis. Mix 2: DNA of the lactic acid bacteria was added at different concentrations: 500 ng Mix 1: DNA of Lactobacillus sakei (DSMZ 6333), Lactococcus lactis (DSMZ 20481), Leuconostoc mesenteroides (DSMZ 20241) and Pediococcus pentosaceus (DSM 20336) at the same concentration (100 ng sakei lane 5, ePCR (mix 1) lane 6, not emulsified control (NEC), non-emulsified control (mix 1) lane 7, conventional PCR (mix 1) lane 8, ePCR (mix 2) lane 9, NEC, non-emulsified control (mix 2) lane 10, conventional PCR (mix 2). Denaturing gradient gel electrophoresis (DGGE) profiles of mixtures of amplicons obtained from DNA extracted from lactic acid bacteria (A) and DNA extracted directly from bulk-collected coagulase-negative catalase-positive cocci (CNCPC) cells. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
